.png)
Cretex Medical Exhibits at SMST 2026 Cretex Medical employees attended the 2026 Shape Memory and Superelastic Technologies (SMST) Conference and Exposition in La Jolla, California. May 12, 2026.
.png)
Cretex Medical Exhibits at SMST 2026 Cretex Medical employees attended the 2026 Shape Memory and Superelastic Technologies (SMST) Conference and Exposition in La Jolla, California. May 12, 2026.

Cretex Medical Exhibits at DeviceTalks Minnesota 2026 Cretex Medical recently attended and exhibited at DeviceTalks Minnesota 2026, connecting with industry leaders and showcasing innovative.
.png)
Cretex Medical Attends ODT Forum 2026 The ODT Forum is hosted by Orthopedic Design and Technology magazine on the campus of the University of Memphis in Tennessee. April 28, 2026 Cretex Medical | QTS.
.png)
Cretex Medical Exhibits at AAOS 2026 AAOS brings together surgeons, specialists, and industry leaders each year for expert-led education, interactive hands-on sessions, and opportunities to explore.
.png)
Cretex Medical Exhibits at MD&M West 2026 Cretex Medical traveled to Anaheim, California to attend and exhibit at MD&M West 2026, one of the largest multi-sector manufacturing trade shows in the.
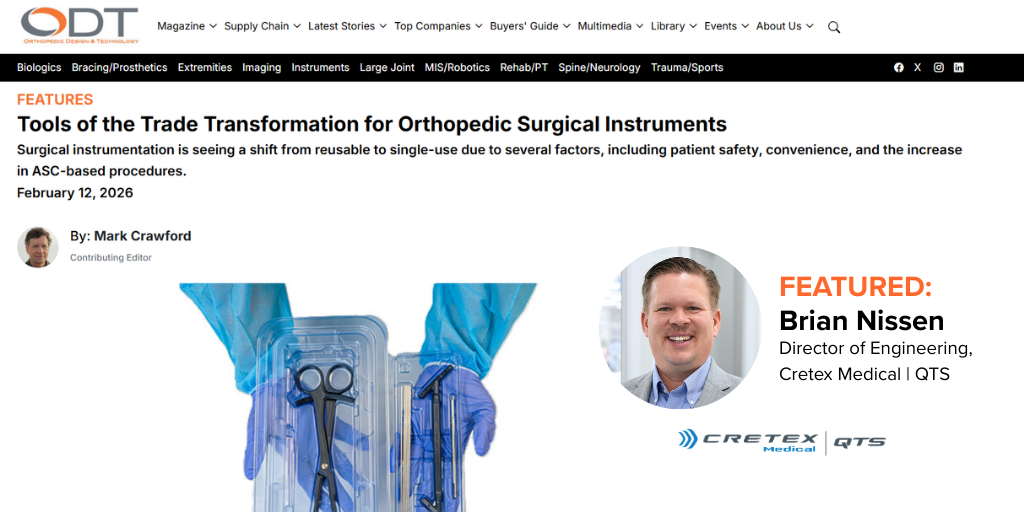
Cretex Medical Featured in ODT Magazine Article on Single-Use Orthopedic Surgical Instruments Brian Nissen, Director of Engineering for Cretex Medical | QTS, was featured in ODT Magazine’s “Tools of.

Cretex Medical | rms Additive Manufacturing (AM) team featured in BONEZONE Magazine Traditional methods are ineffective for testing additively manufactured devices. The AM team at Cretex Medical |.

Cretex Medical Exhibits at MD&M Midwest 2025 Cretex Medical was proud to participate in MD&M Midwest 2025 in Minneapolis, MN, one of the region’s premier gatherings for medical device manufacturers.

Cretex Medical Signs Agreement to Acquire Atemisa Precision, S.A. in Costa Rica Cretex Medical, a leading provider of contract medical device manufacturing and engineering services, announced today.

Cretex Medical Engineering Manager, Emma Danner, featured in MDO Women in Medtech Danner discussed the evolution of the medtech industry, the challenges engineers face, and how to foster a culture of.