
Cretex Medical | QTS Team Members Attend the 2025 Kilmer Conference Cretex Medical | QTS team members Sopheak Srun and Molly Swanson attended AAMI's Kilmer Conference from February 4-6, 2025..

Cretex Medical | QTS Team Members Attend the 2025 Kilmer Conference Cretex Medical | QTS team members Sopheak Srun and Molly Swanson attended AAMI's Kilmer Conference from February 4-6, 2025..
.png)
Cretex Medical Showcases Innovation and Builds Connections at MD&M West 2025 Cretex Medical recently exhibited at MD&M West 2025 from February 4-6. February 10, 2025 Cretex Medical traveled to.

Cretex Medical’s rms Additive Team at ASTM ICAM 2024: Pioneering Additive Manufacturing Innovations In October 2024, Cretex Medical | rms Additive team members Alek Nelson and Ryan Kircher attended.
.png)
Cretex Medical Attends MD&M Minneapolis 2024 Connecting Innovators and Shaping the Future of Medical Technology at MD&M Minneapolis 2024. October 23, 2024 Cretex Medical attends MD&M Minneapolis 2024.

Additive Manufacturing: Moving Beyond Implants - An Orthopedic Innovators Q&A With additive manufacturing becoming more of a production process, it’s critical to find the right suppliers who have.
.png)
Cretex Medical Attends MedTech Ireland 2024 Cretex Medical recently attended MedTech Ireland 2024 in Galway, where we connected with industry leaders, explored cutting-edge medical technologies, and.
Medical Device Machinists Are Multi-Task Masters Medtech machining equipment has evolved to meet customers’ demands for micro components, material variability, more complex shapes, and tighter.
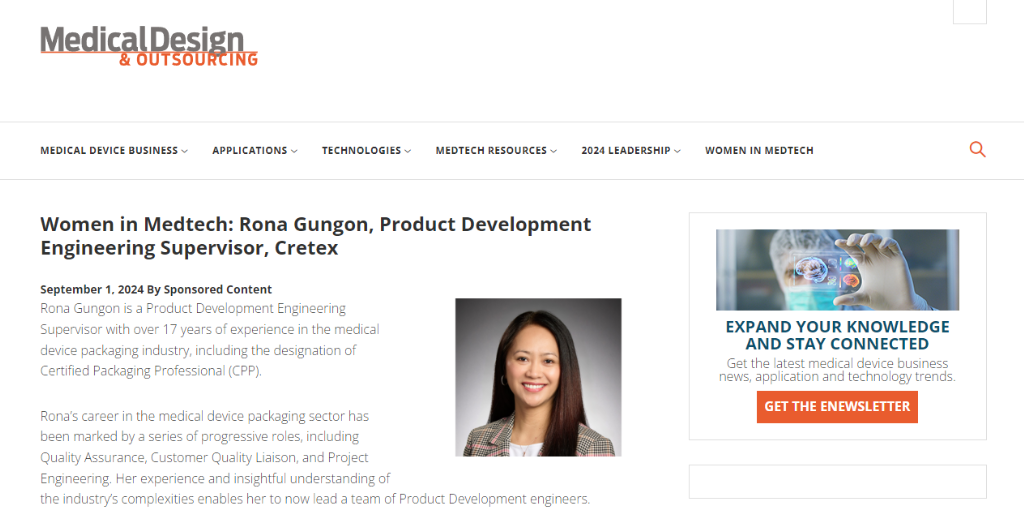
Women in MedTech: Rona Gungon, Product Development Engineering Supervisor at Cretex Medical Rona Gungon is a Product Development Engineering Supervisor with over 17 years of experience in the medical.

Unlock the Potential: Elevate Your Manufacturing with 3D Printing The landscape of healthcare manufacturing is evolving at an unprecedented pace, thanks in part to the impact of additive.

Navigating Current Challenges with Sterilization Capacity Constraints In recent years, the medical device industry has faced growing sterilization capacity constraints. The primary causes are.