
Considerations for Bringing Neurostim Innovations to Market—A Medtech Makers Q&A As alternatives to pharmaceuticals, neuro devices can provide benefits without the side effects for those who require.

Considerations for Bringing Neurostim Innovations to Market—A Medtech Makers Q&A As alternatives to pharmaceuticals, neuro devices can provide benefits without the side effects for those who require.

Sharing Our Additive Manufacturing Expertise with Tech Briefs Readers Additive manufacturing experts at rms recently contributed to an article featured in Tech Briefs’ Special Report: Medical.

Women in Medtech: Janelle Swanson, Business Unit Director at QTS, a Cretex Medical Company by Sponsored Content, Medical Device & Outsourcing | October 3, 2022 Janelle Swanson, Business Unit Director.

Medtech Manufacturing Cleaning and Sterilization Validation—A Medtech Makers Q&A Ensuring components remain residue-free and are properly sterilized are critical tasks for medical device firms. by.

How Industrial CT Scanning Improves Additive Manufacturing OEMs are seeking manufacturing partners who can leverage multiple technologies to design and fabricate the instruments for orthopedic.

One of the most well-known benefits of additive manufacturing (AM) is the ability to integrate elaborate surface technologies. These technologies, such as lattices and gyroids, allow for improved.

Women in Medtech 2021: Angie Hillyard, Spectralytics Engineering Director by Sponsored Content, Medical Device & Outsourcing | October 12, 2021 Angie Hillyard, Engineering Director at Cretex Medical,.

From Powder to Package: Identifying the Right AM Suppliers Medical device contract manufacturer rms Company watched additive manufacturing develop and evolve, and ultimately saw an opportunity to.
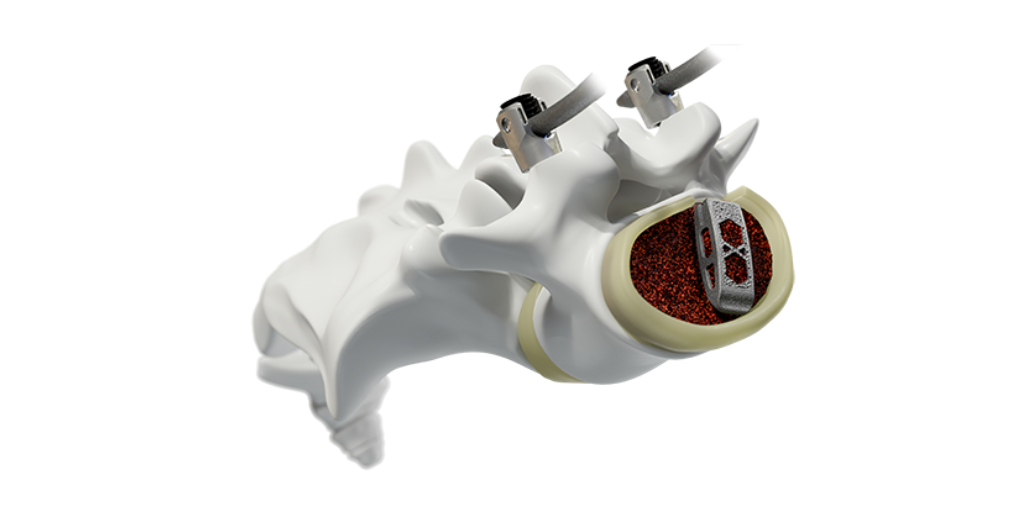
Medtronic Launches Proprietary Surface Technology on AM Devices Medtronic became interested in additive manufacturing (AM) to extend its innovation, unlocking advantages exclusive to the technology..

Our precision metal stamping team (formerly Meier Tool & Engineering) was recently featured in an article by SciTech MN titled Internship Feature: Meier Tool and Engineering. The article focused on.