.png)
Cretex Medical Exhibits at SMST 2026 Cretex Medical employees attended the 2026 Shape Memory and Superelastic Technologies (SMST) Conference and Exposition in La Jolla, California. May 12, 2026.
.png)
Cretex Medical Exhibits at SMST 2026 Cretex Medical employees attended the 2026 Shape Memory and Superelastic Technologies (SMST) Conference and Exposition in La Jolla, California. May 12, 2026.

Cretex Medical Exhibits at DeviceTalks Minnesota 2026 Cretex Medical recently attended and exhibited at DeviceTalks Minnesota 2026, connecting with industry leaders and showcasing innovative.
.png)
Cretex Medical Attends ODT Forum 2026 The ODT Forum is hosted by Orthopedic Design and Technology magazine on the campus of the University of Memphis in Tennessee. April 28, 2026 Cretex Medical | QTS.
.png)
Cretex Medical Exhibits at AAOS 2026 AAOS brings together surgeons, specialists, and industry leaders each year for expert-led education, interactive hands-on sessions, and opportunities to explore.
.png)
Cretex Medical Exhibits at MD&M West 2026 Cretex Medical traveled to Anaheim, California to attend and exhibit at MD&M West 2026, one of the largest multi-sector manufacturing trade shows in the.
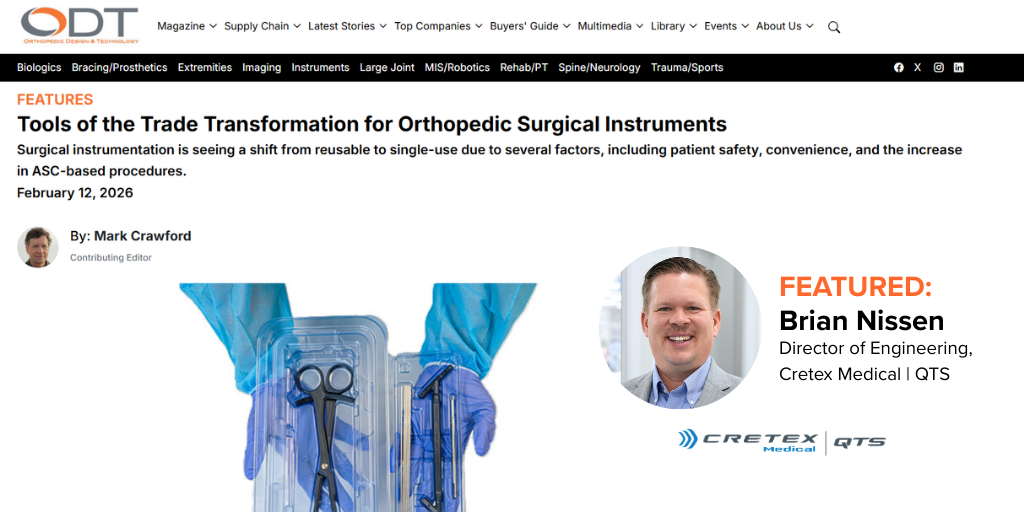
Cretex Medical Featured in ODT Magazine Article on Single-Use Orthopedic Surgical Instruments Brian Nissen, Director of Engineering for Cretex Medical | QTS, was featured in ODT Magazine’s “Tools of.

Cretex Medical | rms Additive Manufacturing (AM) team featured in BONEZONE Magazine Traditional methods are ineffective for testing additively manufactured devices. The AM team at Cretex Medical |.

Cretex Medical Exhibits at MD&M Midwest 2025 Cretex Medical was proud to participate in MD&M Midwest 2025 in Minneapolis, MN, one of the region’s premier gatherings for medical device manufacturers.

Cretex Medical Signs Agreement to Acquire Atemisa Precision, S.A. in Costa Rica Cretex Medical, a leading provider of contract medical device manufacturing and engineering services, announced today.

Cretex Medical Engineering Manager, Emma Danner, featured in MDO Women in Medtech Danner discussed the evolution of the medtech industry, the challenges engineers face, and how to foster a culture of.

Streamlining Orthopedic Device Testing with Product Families Brian Nissen, Director of Engineering at Cretex Medical | QTS, was recently featured in ODT Magazine’s Orthopedic Innovators Q&A series..

Cretex Medical Sponsors Thumbs Up Annual 5K/10K Walk/Run/Bike Event

Cretex Companies Rallies for Communities in Annual Cares-A-Thon Each June, our teams across the country unite for the Cretex Cares-A-Thon, a month-long celebration of service, generosity, and.

Cretex Medical Sponsors & Exhibits at OMTEC 2025 Cretex Medical traveled to Chicago, IL to attend and exhibit at OMTEC 2025, the premier annual event for the orthopedic manufacturing industry,.

Cretex Medical | rms Celebrates Production of One Millionth Additive Manufactured Part Cretex Medical | rms recently celebrated the production of its One Millionth Additive Manufactured Part, a major.
![Cretex Medical | QTS Attends the[PACK]out 2025](https://blog.cretexmedical.com/hubfs/The%5BPACK%5DOut%20Blog%20Cover.png)
Cretex Medical | QTS Attends The[PACK]Out 2025 Two members of the Cretex Medical | QTS team, Rona and Anna, attended the[PACK]out 2025 to explore the latest innovations in sterile packaging. From.

Streamlining Orthopedic Device Testing with Product Families Cretex Medical | QTS Scientists discuss the state of EO and gamma ray sterilization, and how medical device companies can find solutions.

Cretex Medical | rms Receives Minnesota Governor’s Workplace Safety Silver Award Cretex Medical | rms is proud to announce that we were honored with the Minnesota Governor’s Workplace Safety Silver.

Cretex Medical Attends DeviceTalks Boston 2025 DeviceTalks Boston 2025 brought together some of the brightest minds and most forward-thinking companies in the medical device space, and Cretex Medical.
.png)
Cretex Medical | CDT Brooklyn Park Teams Raise $6,950 for Local Charities At Cretex Medical, giving back is a team effort, and our Brooklyn Park employees took that spirit to the next level this.

Cretex Medical Attends MPO MedTech Forum Costa Rica 2025 As Costa Rica’s largest export, the medical device industry is a driving force behind the country’s economy, and a growing focus area for.
.png)
Cretex Medical’s Dave Novak Shares Industry Insights in MPO Magazine Cretex Medical’s Dave Novak was featured in MPO Magazine, where he shared insights on outsourcing trends, strategic growth, and.

Cretex Medical | rms Team Member Ryan Kircher Honored with Distinguished Innovator Operators Award at AMUG 2025 Cretex Medical proudly celebrates Ryan Kircher, Principal Manufacturing Engineer at.

Cretex Medical | rms Presents at the 2025 Additive Manufacturing Users Group (AMUG) Conference The conference is an opportunity for attendees to learn about advancements in additive manufacturing,.
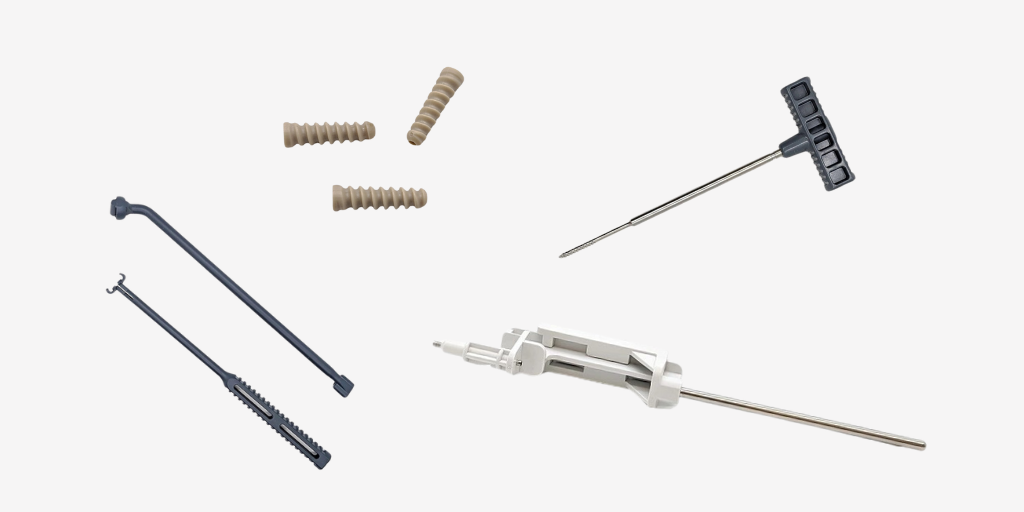
Cretex Medical | CDT’s Kyle Klein Featured in ODT Magazine: Advancing the Future of Molding in Orthopedics Cretex Medical | CDT’s Kyle Klein was featured in ODT Magazine, where he shared how advanced.

Cretex Medical Showcases Expertise at AAOS 2025 Cretex Medical recently exhibited at AAOS 2025 from March 11-13. March 18, 2025 Cretex Medical recently attended the 2025 American Academy of.

Cretex Medical’s Advanced Instrument Capabilities Featured in Orthopedic Design & Technology (ODT) Magazine Korey Lieser, Instruments Operations Manager at Cretex Medical | rms, was featured in.

Cretex Medical | QTS Team Members Attend the 2025 Kilmer Conference Cretex Medical | QTS team members Sopheak Srun and Molly Swanson attended AAMI's Kilmer Conference from February 4-6, 2025..
.png)
Cretex Medical Showcases Innovation and Builds Connections at MD&M West 2025 Cretex Medical recently exhibited at MD&M West 2025 from February 4-6. February 10, 2025 Cretex Medical traveled to.

Cretex Medical’s rms Additive Team at ASTM ICAM 2024: Pioneering Additive Manufacturing Innovations In October 2024, Cretex Medical | rms Additive team members Alek Nelson and Ryan Kircher attended.
.png)
Cretex Medical | rms Volunteers at The Dwelling Place for Domestic Violence Awareness Month October is Domestic Violence Awareness Month - a time to shine a light on the struggles faced by survivors.
.png)
Cretex Medical Attends MD&M Minneapolis 2024 Connecting Innovators and Shaping the Future of Medical Technology at MD&M Minneapolis 2024. October 23, 2024 Cretex Medical attends MD&M Minneapolis 2024.

Additive Manufacturing: Moving Beyond Implants - An Orthopedic Innovators Q&A With additive manufacturing becoming more of a production process, it’s critical to find the right suppliers who have.

Cretex Medical | rms Donates Blankets to the Elderly Cretex Medical | rms employees from Anoka and Coon Rapids crafted 12 blankets and over a dozen cards for elderly residents, with volunteer.
.png)
Cretex Medical Attends MedTech Ireland 2024 Cretex Medical recently attended MedTech Ireland 2024 in Galway, where we connected with industry leaders, explored cutting-edge medical technologies, and.

Cretex Medical Shows Continuous Support by Participating in Thumbs Up’s 11th Annual 5K & 10K Run/Walk/Bike As a long-time supporter of Thumbs Up, Cretex Medical showed up with a strong team of.

Corporate Social Responsibility (CSR) at Cretex Companies At Cretex, our values are the foundation of everything we do. While times may evolve and new challenges emerge, our unwavering commitment to.
Medical Device Machinists Are Multi-Task Masters Medtech machining equipment has evolved to meet customers’ demands for micro components, material variability, more complex shapes, and tighter.
Women in MedTech: Rona Gungon, Product Development Engineering Supervisor at Cretex Medical Rona Gungon is a Product Development Engineering Supervisor with over 17 years of experience in the medical.

Cretex Medical Employees Unite for the Community: A Successful Backpack Drive Cretex Medical recently hosted an Adopt-A-Backpack Backpack Drive for the Anoka-Hennepin School District.

A Sunny Day of Fun and Giving Back: Cretex Companies' Annual Golf Tournament Thanks to everyone's generosity, we raised $115 and donated 367 pounds of food to the ACBC Food Shelf, making a meaningful.

Unlock the Potential: Elevate Your Manufacturing with 3D Printing The landscape of healthcare manufacturing is evolving at an unprecedented pace, thanks in part to the impact of additive.
.png)
Cretex Medical Announces Plans for Costa Rica Facility Cretex Medical announced today that they have broken ground on a new production facility in Cartago, Costa Rica. The 65,000-square-foot medical.

Navigating Current Challenges with Sterilization Capacity Constraints In recent years, the medical device industry has faced growing sterilization capacity constraints. The primary causes are.

Cretex Medical Shines at OMTEC 2024 "This event highlights Cretex Medical's ability to unify our capabilities to create elegant solutions for our customers' most challenging issues. It's wonderful to.

Technology Trends for Structural Heart Solutions — A Medtech Makers Q&A Replacing heart valves has become a more successful medical procedure thanks to the devices used to perform it. by Sean Fenske,.

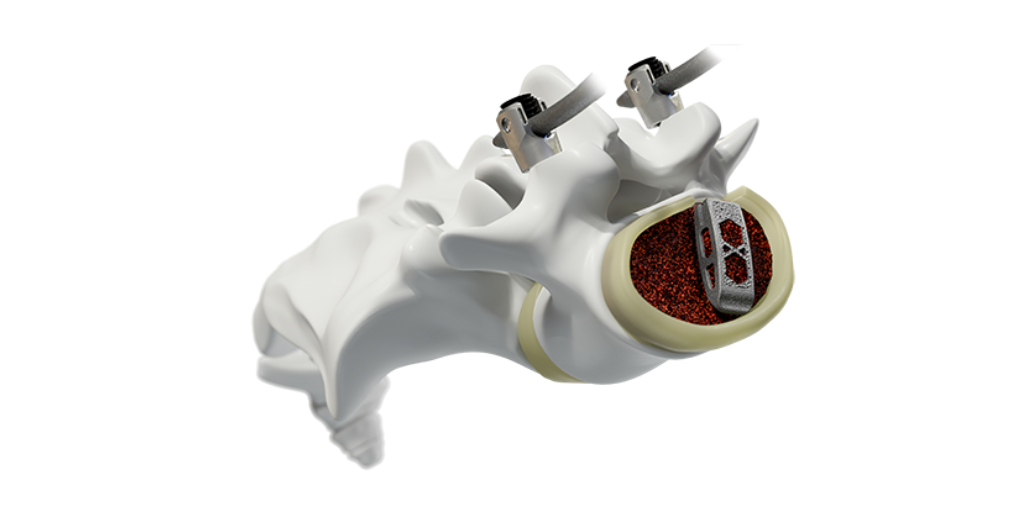
Furthering Orthopedic Implant Fabrication Methods Developers and manufacturers of implants have added new tools to their belts to create orthopedic technologies using innovative techniques. by Mark.
![Emily Graba and Rona Gungon attend the[PACK]out 2024](https://blog.cretexmedical.com/hubfs/the%5BPACK%5Dout.png)
Emily Graba and Rona Gungon attend the[PACK]out 2024 In May, 2024, QTS employees Emily Graba, Process Development Engineer, and Rona Gungon, Product Development Engineering Supervisor, attended.

Nick Morrison Featured in ODT Forum 2024 Cretex Companies COO Nick Morrison participated in the session OEM/CMO Relationship Outlook: The Impact of Consolidation. April 24, 2024 Cretex Medical COO.

Cretex Companies, Inc. Plastic Recycling Program Secures Bench for Local Elementary School In 2023, Cretex employees at the Brooklyn Park facility began participating in a program that recycled film.
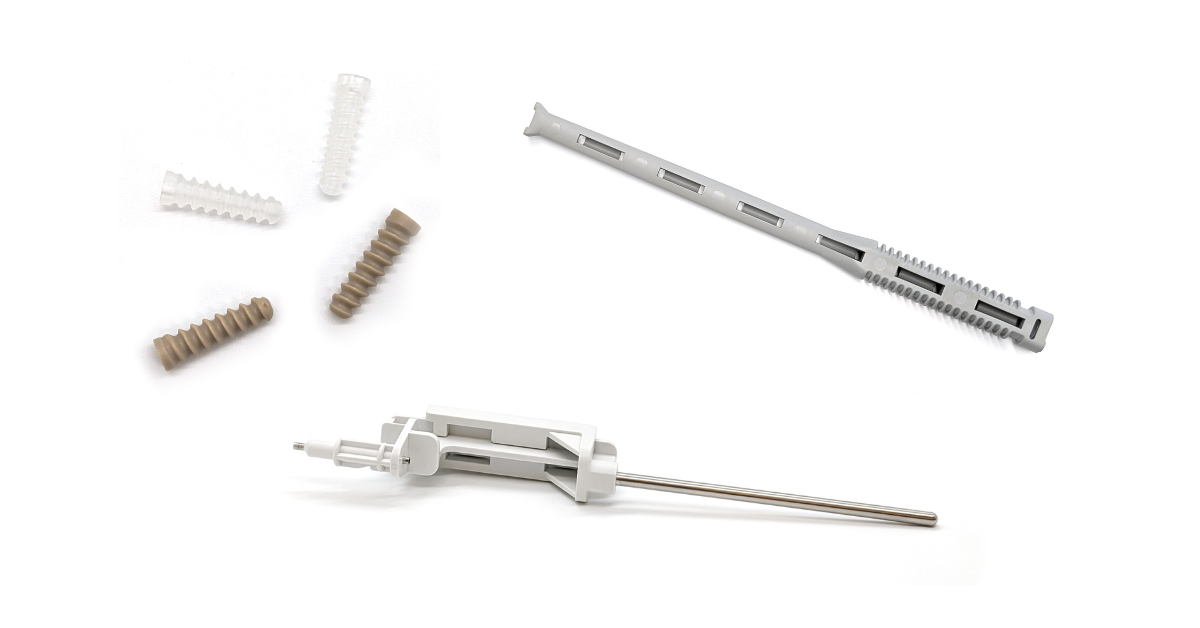
Molding Is Taking Its Place in Orthopedic Device Manufacturing As molders continue to expand capabilities attractive to orthopedic device makers, the fabrication process is growing in use. by Mark.

Abbott Electrophysiology Leaders Highlight Current Portfolio, Give Glimpse of Potential PFA Device In the latest episode of AbbottTalks, Abbott Cardiovascular’s leaders, Christopher Piorkowski, MD,.

QTS Renovation: A Major Transformation for Future Success In 2023, Cretex Medical | Quality Tech Services (QTS) embarked on a remarkable journey of transformation with the completion of a.
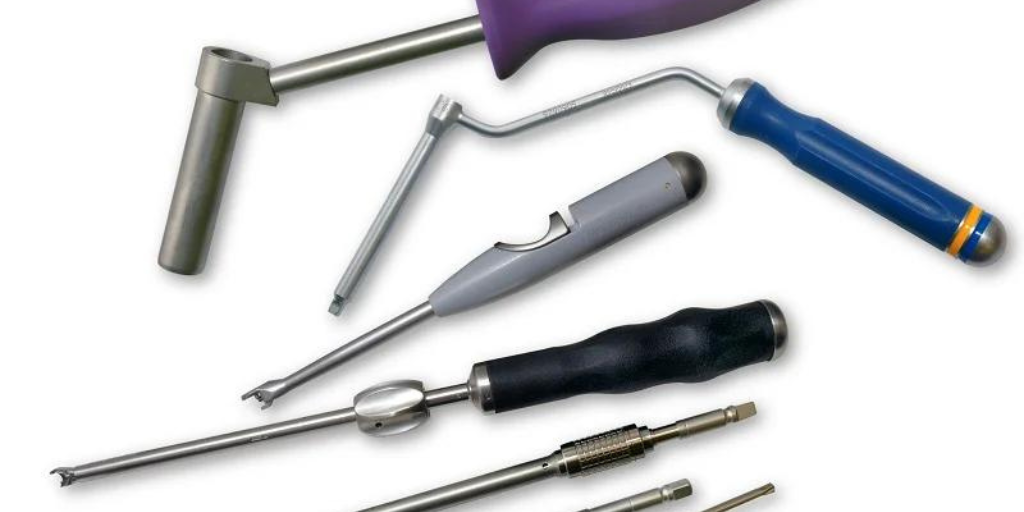
Cretex Medical’s Approach to Surgical Instrumentation Featured in Orthopedic Design & Technology (ODT) Magazine Granger Lubich, Engineering Manager for Cretex Medical | rms was featured in Orthopedic.
Overkilling Uncertainty, New TIR Seeks Clarity for Interpreting Bioburden Data When companies are testing a medical device’s bioburden — a measure of how much and what types of viable organisms are.

Leveraging Automation to Improve Inspection Quality—An Orthopedic Innovators Q&A Automation not only addresses labor shortage concerns, but also enhances the overall output by improving the.

Considerations for Bringing Neurostim Innovations to Market—A Medtech Makers Q&A As alternatives to pharmaceuticals, neuro devices can provide benefits without the side effects for those who require.

February 8, 2023 is Sterile Packaging Day. The day was started by the SPMC (Sterile Packaging Manufacturers Council) to honor and thank their supply chain partners. This year’s theme is “Designed to.

What holiday could be more perfect to celebrate at QTS than Sterile Packaging Day? This year, the Sterile Packaging Manufacturers Council (SPMC) declared Wednesday, March 10 to be the first ever.

Sharing Our Additive Manufacturing Expertise with Tech Briefs Readers Additive manufacturing experts at rms recently contributed to an article featured in Tech Briefs’ Special Report: Medical.

Women in Medtech: Janelle Swanson, Business Unit Director at QTS, a Cretex Medical Company by Sponsored Content, Medical Device & Outsourcing | October 3, 2022 Janelle Swanson, Business Unit Director.

Medtech Manufacturing Cleaning and Sterilization Validation—A Medtech Makers Q&A Ensuring components remain residue-free and are properly sterilized are critical tasks for medical device firms. by.

How Industrial CT Scanning Improves Additive Manufacturing OEMs are seeking manufacturing partners who can leverage multiple technologies to design and fabricate the instruments for orthopedic.

Abbott Electrophysiology Leaders Highlight Current Portfolio, Give Glimpse of Potential PFA Device In the latest episode of AbbottTalks, Abbott Cardiovascular’s leaders, Christopher Piorkowski, MD,.

One of the most well-known benefits of additive manufacturing (AM) is the ability to integrate elaborate surface technologies. These technologies, such as lattices and gyroids, allow for improved.

Spectralytics - Impacting Lives Daily Spectralytics has been a pioneer in laser technology for the medical device space for over twenty years, paving the way for current industry standards. by Laure.

AM for Production: Where Conventional Lessons Do and Do Not Apply As RMS has grown its additive manufacturing division, it has discovered which principles of subtractive manufacturing apply to.

New Sterility Assurance Strategies for Products Sensitive to Sterilization Processes QTS was featured in AAMI ARRAY by Sopheak Srun & Molly Swanson | October 26, 2021 Sopheak Srun, MPH, SM(NRCM),.

Cleaning Considerations for Additively Manufactured Parts Cretex Medical | QTS was recently featured in BONEZONE magazine to discuss additive manufacturing.

Women in Medtech 2021: Angie Hillyard, Spectralytics Engineering Director by Sponsored Content, Medical Device & Outsourcing | October 12, 2021 Angie Hillyard, Engineering Director at Cretex Medical,.
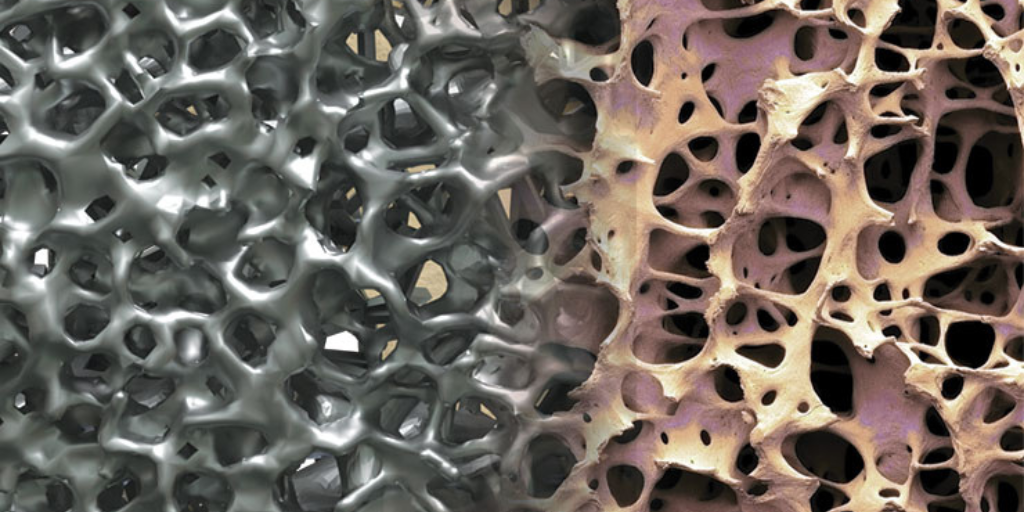
Metal Injection Molding and Metal 3D Printing Examination Both MIM and metal AM serve as alternative options to machining for medtech manufacturers requiring metal components.
QTS Collaborates with Other Industry Experts to Publish Best Practices Article on Tests of Sterility for EO Sterilization Sopheak Srun, a principal sterilization specialist at QTS, recently worked.

The laser team in Dassel had a great time supporting their four-legged friends in the community! Together, they were able to donate 1,500 items to Hawk Creek Animal Shelter. Big thanks to everyone.

Sopheak Srun Appointed Co-Chair of an AAMI Working Group Sopheak, QTS Principal Sterilization Specialist, will be contributing to new and existing ISO standards to be upheld internationally.

Cretex Companies Announces Plans for New Facility Cretex Companies have signed a purchase agreement for land in Brooklyn Park, MN for a brand new facility.

From Powder to Package: Identifying the Right AM Suppliers Medical device contract manufacturer rms Company watched additive manufacturing develop and evolve, and ultimately saw an opportunity to.

Cretex Companies Announces New CFO Cretex Companies has announced the retirement of CFO Steve Ragaller.

Cretex Companies Announces Leadership Transition Cretex Companies has announced the retirement of CEO Lynn Schuler
Medtronic Launches Proprietary Surface Technology on AM Devices Medtronic became interested in additive manufacturing (AM) to extend its innovation, unlocking advantages exclusive to the technology..

Cretex Medical | CDT Company, JunoPacific, Announces Operations Consolidation JunoPacific is in the process of combining its Redwood City and Soquel operations into a single site in Soquel, CA.

QTS receives Global First in Sterile Device Packaging from MedAccred Cretex Medical | QTS, a leader in medical device outsourcing solutions, has received accreditation from MedAccred® as the Global.

Each year, QTS hosts a holiday party to celebrate the festive season and to recognize the achievements and hard work of our employees. The evening began with our annual awards ceremony, highlighting.

Our precision metal stamping team (formerly Meier Tool & Engineering) was recently featured in an article by SciTech MN titled Internship Feature: Meier Tool and Engineering. The article focused on.

Meier Celebrates 40 Years As Meier celebrates its 40th-anniversary milestone, its focus—as always—is on its customers as it continues to grow, innovate and invest to meet their ever-expanding needs..

rms to Expand its Coon Rapids Facility rms has announced that it will be adding 60,000 sq. feet to its Coon Rapids manufacturing facility with construction beginning in 2019.

On July 18, QTS decked out the beautiful Hyland Hills Chalet with totally rad 80’s decor, and many attendees also dressed the part! The warm, sunny day allowed for employees and their family members.

On Saturday, July 13, a few of our employees geared up for the 5th Annual Cretex Companies Golf Tournament. This event was a great opportunity for employees throughout our family of companies to get.

In June of 2019, QTS Principal Sterilization Specialist Sopheak Srun attended the 10th Annual Kilmer Conference in Dublin, Ireland. The Kilmer Conference is an invite-only conference hosted by.

Here at QTS, we have an ongoing initiative to give back to our community. In May, we organized and sponsored our annual Dresser Build event at Bridging, where a group of our staff members volunteered.

Expanding to Meet Your Needs: QTS Opens New Warehouse QTS is pleased to announce the consolidation of our warehouse operations into a single, new location. August 26, 2018 Cretex Medical | QTS.

QTS held our Annual Company Picnic in late July and everyone had a blast! Many of our employees attended, and despite the bothersome weather, everyone was still able to enjoy all the festivities. In.

Cretex Companies Acquires Leading Medical Packaging Services Provider QTS is widely recognized as a leading provider of validated assembly, kitting, and packaging solutions. June 13, 2017 Cretex.

Sopheak Srun, QTS Principal Sterilization Specialist

Compliance to the UDI regulation includes the FDA public database, GUDID. The first phase of the Global Unique Device Identification Database (GUDID): Guidance for Industry document is now available..

DuPont Tyvek Transition Update DuPont is transitioning Tyvek® 1073B and Tyvek® 1059B to manufacturing lines that use the latest flash-spinning technology to help ensure greater continuity and.

Unique Device Identification (UDI) Final Rule was announced by the FDA on September 20, 2013. The rule was published in the Code of Federal Regulations as of September 24, 2013. Implementation for.

QTS staff, Sopheak Srun and Brian Nissen, co-authored a research paper regarding Medical Device SALs and Surgical Site Infections. The paper was originally published in the May/June 2012 issue in.